Medical Use
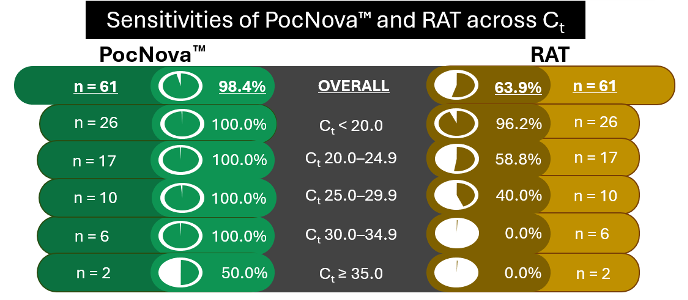
In a clinical trial conducted at Queen Mary Hospital's Accident and Emergency Department for COVID-19
testing during July–September 2022, PocNova™ achieved accuracy, sensitivity, and specificity of 99.6%,
98.4%, and 100.0%, respectively, with reference to real-time PCR.

Non-Medical Use
PocNAT™ has also conducted food and environmental test trials with SGS Hong Kong, Maxim's Group, Hotel ICON,
and International Gourmet Foods since 2023 Q3 in Hong Kong. PocNova™ has achieved equivalent accuracy with
reference to centralized laboratory results based on PCR (8–48 hours) and culture (3–14 days) but much
faster (< 30 minutes). Since November 2024, on-site test trials collaborating with AMAE and a state-owned
laboratory in Shenzhen China achieved consistent test results for a total of more than 300 tests for real
environmental swabs and food samples collected during large public events.

Collaborators






